Compiled by Jørn Tore Haugen,
Last revised: January 26th, 2026
Studies of ME patients using two-day CPET (Cardiopulmonary Exercise Test) show that the disease is not caused by exercise phobias, deconditioning or ‘illness beliefs’. Measurements on day two versus day one show at anaerobic threshold, reduced oxygen uptake (VO2) and work capacity, and increased heart rate. At same workload, the lactate concentration increases at day two. Abnormalities in the brain are seen. This is entirely different to other disease we know including cardiovascular disease, lung disease, end-stage renal disease, hypertension, cystic fibrosis and multiple sclerosis.
CPET provides an explanation for why ME patients can ‘crash’ even after modest exertion. The reduced aerobic capacity of ME patients results in a switch to anaerobic metabolism and patients can ‘crash’ if they over-exert resulting in a post exertional malaise (PEM). Exercise that triggers PEM in ME patients leads to a sustained immune inflammatory response that is distinct from what seen in sedentary or deconditioned people.
Consequently, two-day CPET can be considered as an objective marker for PEM in ME patients due to measurable deficits in metabolism.
PESE: postexertional symptom exacerbation ≈ PEM: post exertional malaise.
Guidelines
The American College of Sports Medicine’s Guidelines for Exercise Testing and Prescription, for the first time, includes a section on ME/CFS. October 2025: ACSM’s Guidelines for Exercise Testing and Prescription, 12th edition
ACSM is a leading authority on exercise science, and these guidelines serve as a foundational text for exercise professionals to guide safe and effective exercise testing and programming. The guidelines were released on the use of cardiopulmonary exercise testing (CPET) in research and clinical practice for myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). These guidelines are important because CPET has long been one of the few tools that objectively capture the hallmark feature of ME/CFS: post-exertional malaise (PEM). Bateman Horne Center: New CPET guidelines: What they mean for people with ME/CFS.
Study references
49. ME/CFS and Long COVID Demonstrate Similar Bioenergetic Impairment and Recovery Failure on Two-Day Cardiopulmonary Exercise TestingTodd Davenport, Staci Stevens, Jared Stevens, Mark Van Ness Clinical & Translational Metabolism, Preprint (Version 1), 22 January 2026, https://doi.org/10.21203/rs.3.rs-8606329/v1
Conclusion/Main finding: “There were significant reductions in oxygen consumption (VO₂) and workload at the ventilatory anaerobic threshold (VAT) in both patient groups compared to non-disabled controls, with larger effect sizes at VAT than at peak exertion. Performance decrements were observed in both sexes. Females exhibited more pronounced abnormalities and significant group by test effects. No significant differences were observed between patient groups. Severe disability based on impaired VO₂ was prevalent in both patient groups. Hemodynamic and ventilatory measures were within normal ranges. ME/CFS and Long Covid both involve a functionally significant bioenergetic failure complicated by inadequate post-exertional recovery, which is similar between the conditions and unexplained by hemodynamic and ventilatory changes. Findings support the utility of two-day CPET as an objective measure of PEM and functional impairment.”
Inclusion criteria: Unknown
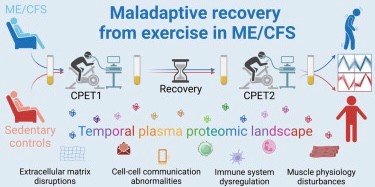
48. Temporal dynamics of the plasma proteomic landscape reveals maladaptation in ME/CFS following exertion Germain, Arnaud et al. Molecular & Cellular Proteomics, Volume 0, Issue 0, 101467, 2025 https://www.mcponline.org/article/S1535-9476(25)00566-3/fulltext
Conclusion/Main finding: «Key findings included suppression of T and B cell signaling, downregulation of IL-17 and cell-cell communication pathways, and upregulation of glycolysis/gluconeogenesis, suggestive of mitochondrial stress and impaired immune recovery from exercise. Proteomic associations with physiological performance (VO2max, anaerobic threshold) revealed disruptions between protein abundance and exercise capacity in ME/CFS versus controls. Correlations with symptom severity linked changes in immune-related proteins and ME/CFS symptoms including muscle pain, recurrent sore throat, and lymph node tenderness. Sex-stratified analyses revealed distinct molecular responses between females and males, emphasizing the importance of considering sex as a biological variable in ME/CFS research. Finally, our analysis of sedentary controls contributes new data of molecular responses to acute exertion in a predominantly female sedentary cohort, a population historically underrepresented in exercise physiology studies. Together, these findings underscore the value of dynamic, proteomic profiling over time for characterizing maladaptive responses to exertion in ME/CFS and provide a foundation for deeper mechanistic investigation into PEM.»
Inclusion criteria: Canada Consensus Criteria
..
47. Blood Flow To The Head Is Reduced In A Patient With Myalgic Encephalomyelitis With Confirmed Post-exertional Malaise Lee, D, Stevens, S, Davenport, T, Stevens, J, Dowell, T, VanNess, M.: 2417. Medicine & Science in Sports & Exercise 56(10S):p 894-895, October 2024. | DOI: 10.1249/01.mss.0001060360.80310.99 https://journals.lww.com/acsm-msse/fulltext/2024/10001/blood_flow_to_the_head_is_reduced_in_a_patient.2013.aspx
Conclusion/Main finding: «Reduced blood flow to the head during PEM may partially explain ME symptomatology. The novel in-ear device shows promise as a diagnostic and biofeedback tool, warranting further research in larger studies.»
Inclusion criteria: PEM
46. Exercise-induced Changes in Microclotting and Cytokine Levels Point to Vascular Injury and Inflammation in People with Long COVID, Callum Thomas, Massimo Nunes, Jan H. Pretorius et al. 26 May 2025, PREPRINT (Version 1) https://doi.org/10.21203/rs.3.rs-6717727/v1
Conclusion/Main finding: «This study provides the first evidence of a biological basis that might explain exercise-induced symptom exacerbation in people with Long COVID through microclot fragmentation, which may contribute to systemic inflammation. This has important implications for Long COVID rehabilitation practices that seek to improve health outcomes through exercise therapies that may have the capacity to be harmful for people living with Long COVID and underscores the need for targeted therapeutic strategies that consider microclot clearance and endothelial repair.».
Inclusion criteria: A confirmed history of SARS-CoV-2 infection, a clinical diagnosis of Long COVID and the presence of PESE with a low risk of experiencing PEM.
45. Towards an understanding of physical activity-induced post-exertional malaise: Insights into microvascular alterations and immunometabolic interactions in post-COVID condition and myalgic encephalomyelitis/chronic fatigue syndrome. Haunhorst, S., Dudziak, D., Scheibenbogen, C. et al. Infection 53, 1–13 (2025). https://doi.org/10.1007/s15010-024-02386-8
Conclusion/Main finding: «Upon physical activity, affected patients exhibit a reduced systemic oxygen extraction and oxidative phosphorylation capacity. Accumulating evidence suggests that these are mediated by dysfunctions in mitochondrial capacities and microcirculation that are maintained by latent immune activation, conjointly impairing peripheral bioenergetics. Aggravating deficits in tissue perfusion and oxygen utilization during activities cause exertional intolerance that are frequently accompanied by tachycardia, dyspnea, early cessation of activity and elicit downstream metabolic effects. The accumulation of molecules such as lactate, reactive oxygen species or prostaglandins might trigger local and systemic immune activation. Subsequent intensification of bioenergetic inflexibilities, muscular ionic disturbances and modulation of central nervous system functions can lead to an exacerbation of existing pathologies and symptoms.».
Inclusion criteria: N/A, literature review
44. Cardiopulmonary and metabolic responses during a 2-day CPET in myalgic encephalomyelitis/chronic fatigue syndrome: translating reduced oxygen consumption to impairment status to treatment considerations. Keller, B., Receno, C.N., Franconi, C.J. et al. J Transl Med 22, 627 (2024). https://doi.org/10.1186/s12967-024-05410-5
Conclusion/Main finding: «Unlike CTL, ME/CFS failed to reproduce CPET-1 measures during CPET-2 with significant declines at peak exertion in work, exercise time, e, O2, CO2, T, HR, O2pulse, DBP, and RPP. Likewise, CPET-2 declines were observed at VAT for e/CO2, PetCO2, O2pulse, work, O2 and SBP. Perception of effort (RPE) exceeded maximum effort criteria for ME/CFS and CTL on both CPETs. Results were similar in matched pairs. Intraclass correlations revealed greater stability in CPET variables across test days in CTL compared to ME/CFS owing to CPET-2 declines in ME/CFS. Lastly, CPET-2 data signaled more severe impairment status for ME/CFS compared to CPET-1.
Presently, this is the largest 2-d CPET study of ME/CFS to substantiate impaired recovery in ME/CFS following an exertional stressor. Abnormal post-exertional CPET responses persisted compared to CTL matched for aerobic capacity, indicating that fitness level does not predispose to exertion intolerance in ME/CFS. Moreover, contributions to exertion intolerance in ME/CFS by disrupted cardiac, pulmonary, and metabolic factors implicates autonomic nervous system dysregulation of blood flow and oxygen delivery for energy metabolism. The observable declines in post-exertional energy metabolism translate notably to a worsening of impairment status. Treatment considerations to address tangible reductions in physiological function are proffered.».
Inclusion criteria: Canadian Consensus Criteria
43. Single-cell transcriptomics of the immune system in ME/CFS at baseline and following symptom provocation, Luyen Tien Vu, Faraz Ahmed, Hongya Zhu, David Shing Huk Iu, Elizabeth A. Fogarty, Yeonui Kwak, Weizhong Chen, Carl J. Franconi, Paul R. Munn, Ann E. Tate, Susan M. Levine, Jared Stevens, Xiangling Mao, Dikoma C. Shungu, Geoffrey E. Moore, Betsy A. Keller, Maureen R. Hanson, Jennifer K. Grenier, Andrew Grimson. Cell Reports Medicine VOLUME 5, ISSUE 1, 101373, JANUARY 16, 2024 DOI: https://doi.org/10.1016/j.xcrm.2023.101373
Conclusion/Main finding: «Comparing the transcriptome at baseline and postexercise challenge, we discover patterns indicative of improper platelet activation in patients, with minimal changes elsewhere in the immune system. Taken together, these data identify immunological defects present at baseline in patients and an additional layer of dysregulation in platelets.».
Inclusion criteria: PEM. SEID criteria (acc. to IOM report 2015)
42. A Unique Circular RNA Expression Pattern in the Peripheral Blood of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Patients, Yuning Cheng, Si-Mei Xu, Konii Takenaka, Grace Lindner, Ashton Curry-Hyde, Michael Janitz Gene, 2023, 147568, ISSN 0378-1119, https://doi.org/10.1016/j.gene.2023.147568
Conclusion/Main finding: «This report comprises the first study on circRNA profile in ME/CFS. Here, expression profiling of circRNAs in ME/CFS patients and healthy individuals who underwent two CPETs over a seven-day period was analysed. We identified specific circRNAs showing differential expression at different time points, before and after exercise, as well as enriched GO terms implicated by the host genes of uniquely expressed circRNAs in ME/CFS patients.».
Inclusion criteria: Unknown.
41. Exercise Pathophysiology in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome and Post-Acute Sequelae of SARS-CoV-2: More in Common Than Not? Phillip Joseph, Inderjit Singh, Rudolf Oliveira, Christine A. Capone, Mary P. Mullen, Dane B. Cook, Mary Catherine Stovall, Johanna Squires, Kristine Madsen, Aaron B. Waxman, David M. Systrom, CHEST 5617, S0012-3692(23)00502-0, DOI: 10.1016/j.chest.2023.03.049
Conclusion/Main finding: «PASC and ME/CFS overlap in both symptom burden and exercise derangements. Noninvasive CPET is useful in characterizing aerobic capacity and evaluating ventilatory inefficiency, the latter caused by hyperventilation. Two-day noninvasive CPET protocols may provide a diagnostic tool by showing a decrement in peak VO2 on day two, potentially due to PEM. Neurovascular dysregulation observed with invasive CPET further explains exercise intolerance in PASC and ME/CFS through impaired cardiac preload and peripheral oxygen extraction, associated with autonomic dysfunction, small fiber neuropathy, ganglionopathy, and mitochondrial dysfunction. Future studies targeting these pathways are needed to reduce the substantial global burden of PASC and ME/CFS.».
Inclusion criteria: SEID criteria (acc. to IOM report 2015)
40. Two Symptoms Can Accurately Identify Post-exertional Malaise in Myalgic Encephalomyelitis/chronic Fatigue Syndrome. Davenport, Todd E. et al. 1 Jan. 2023 : 1 – 15. DOI: 10.3233/WOR-220554
Conclusion/Main finding: «Although PEM is a complex phenomenon, researchers and clinicians may not have to engage in lengthy conversations or utilize complicated questionnaires to identify its existence. Medical professionals can efficiently assess for PEM by focusing on a specific set of post-exertional symptoms and the overall functional impact of those symptoms in the days following physical exertion.»
Inclusion criteria: Fukuda 1994 criteria (CDC) and self-reported PEM or had been diagnosed by their physician with ME/CFS.
39. Recovery from Exercise in Persons with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS). Moore GE, Keller BA, Stevens J, Mao X, Stevens SR, Chia JK, Levine SM, Franconi CJ, Hanson MR. Medicina. 2023; 59(3):571. https://doi.org/10.3390/medicina59030571
Conclusion/Main finding: «ME/CFS subjects took an average of about two weeks to recover from a 2-day CPET, whereas sedentary controls needed only two days. These data quantitate the prolonged recovery time in ME/CFS and improve the ability to obtain well-informed consent prior to doing exercise testing in persons with ME/CFS. Quantitative monitoring of PEM symptoms may provide a method to help manage PEM.»
Inclusion criteria: Canada Consensus Criteria
38. A 2-day cardiopulmonary exercise test in chronic fatigue syndrome patients who were exposed to humidifier disinfectants, Jong-Han Leem, Hyoung-Eun Jeon, Hun Nam, Hwan-Cheol Kim, Kyung-Lim Joa, Environmental Analysis Health and Toxicology Vol: 37(4), Article ID: e2022033, 7 pages https://doi.org/10.5620/eaht.2022033 eISSN: 2671-9525. 2022. https://eaht.org/upload/pdf/eaht-37-4-e2022033.pdf
Conclusion/Main finding: «In the 2-day CPET, the peak oxygen consumption (VO2peak), VO2 at ventilatory threshold (VO2@VT), time to reach VO2peak, and time to reach VO2@VT were significantly decreased (p<0.001). The peak O2 pulse and O2 pulse at VT also decreased significantly (p<0.001). A 6-minute walk test revealed significantly decreased distance (p<0.01). … Therefore, a 2-day CPET is an objective measure to differentiate fatigue conditions in people with CFS symptoms who have been exposed to HDs (humidifier disinfectants).»
Inclusion criteria: Fukuda/CDC
37. Neurovascular Dysregulation and Acute Exercise Intolerance in ME/CFS A Randomized, Placebo-Controlled Trial of Pyridostigmine. Phillip Joseph, MD; Rosa Pari, MD; Sarah Miller, BS; Arabella Warren, BS; Mary Catherine Stovall, BS; Johanna Squires, MSc; Chia-Jung Chang, PhD; Wenzhong Xiao, PhD; Aaron B. Waxman, MD, PhD; and David M. Systrom, MD, American College of Chest Physicians. Published by Elsevier Inc. DOI: 10.1016/j.chest.2022.04.146 Published: May 05, 2022, https://journal.chestnet.org/article/S0012-3692(22)00890-X/fulltext
Conclusion/Main finding: «Pyridostigmine improves peak VO2 in ME/CFS by increasing cardiac output and right ventricular filling pressures. Worsening peak exercise VO2, cardiac output, and right atrial pressure following placebo may signal the onset of postexertional malaise. We suggest that treatable neurovascular dysregulation underlies acute exercise intolerance in ME/CFS.»
Inclusion criteria: SEID criteria (acc. to IOM report 2015)
36. Repeated maximal exercise tests of peak oxygen consumption in people with myalgic encephalomyelitis/chronic fatigue syndrom: a systematic review and meta-analysis. John Derek Franklin & Michael Graham (2022) Published online: 16 August 2022, Fatigue: Biomedicine, Health & Behavior, DOI: 10.1080/21641846.2022.2108628
Conclusion/Main finding: «Synthesised data indicate that people with ME/CFS demonstrate a clinically significant test–retest reduction in work rate at the anaerobic threshold when compared to apparently healthy controls.».
Inclusion criteria: Metaanalysis
35. Plasma metabolomics reveals disrupted response and recovery following maximal exercise in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Arnaud Germain, Ludovic Giloteaux, Geoffrey E. Moore, Susan M. Levine, John K. Chia, Betsy A. Keller, Jared Stevens, Carl J. Franconi, Xiangling Mao, Dikoma C. Shungu, Andrew Grimson, and Maureen R. Hanson, JCI Insight. 2022. DOI: 10.1172/jci.insight.157621
Conclusion/Main finding: «The 24-hour recovery period was distinct in the ME/CFS cohort, with over a quarter of the identified pathways statistically different. The pathways that are uniquely different 24 hours after an exercise challenge provide clues to metabolic disruptions that lead to PEM. Numerous altered pathways were observed to depend on glutamate metabolism, a crucial component to the homeostasis of many organs in the body, including the brain.»
Inclusion criteria: Canada Consensus Criteria
34. Differential Effects of Exercise on fMRI of the Midbrain Ascending Arousal Network Nuclei in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) and Gulf War Illness (GWI) in a Model of Postexertional Malaise (PEM). Baraniuk JN, Amar A, Pepermitwala H, Washington SD. Brain Sciences. 2022; 12(1):78. https://doi.org/10.3390/brainsci12010078
Conclusion/Main finding: «Exercise caused the opposite effects with increased activation in ME/CFS but decreased activation in GWI, indicating different pathophysiological responses to exertion and mechanisms of disease. Midbrain and isthmus nuclei contribute to postexertional malaise in ME/CFS and GWI.».
Inclusion criteria: Fukuda and Canada Consensus Criteria
33. Review of the Midbrain Ascending Arousal Network Nuclei and Implications for Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS), Gulf War Illness (GWI) and Postexertional Malaise (PEM) James N. Baraniuk, Department of Medicine, Georgetown University, Washington, DC 20057, USA Academic Editor: Luigi De Gennaro Brain Sci. 2022, 12(2), 132; https://doi.org/10.3390/brainsci12020132
Conclusion/Main finding: «Prior to exercise, ME/CFS had generally lower blood oxygenation level dependent (BOLD) signals than controls. However, after exercise, ME/CFS had elevated activation of the anterior node of the DMN in the medial prefrontal cortex compared to pre-exercise and to controls»
Inclusion criteria: Unknown. CDC, CCC, SEID and NICE-2020 draft guidelines are discussed.
32. Markers of Cardiac Autonomic Function During Consecutive Day Peak Exercise Tests in People With Myalgic Encephalomyelitis/Chronic Fatigue Syndrome, Maximillian J. Nelson1, Jonathan D. Buckley, Rebecca L. Thomson, Clint R. Bellenger, Kade Davison, Front. Physiol., 14 December 2021 Sec. Exercise Physiology Volume 12 – 2021, DOI: 10.3389/fphys.2021.771899
Conclusion/Main finding: «Heart rate markers of autonomic function were unchanged in ME/CFS patients in the presence of post-exertional malaise, induced by maximal CPET on consecutive days. HR parameters assessed during this protocol are unlikely to represent a useful biomarker of the condition.».
Inclusion criteria: CDC, Canada Consensus Criteria or International Consensus Criteria.
31. Submaximal Exercise Provokes Increased Activation of the Anterior Default Mode Network During the Resting State as a Biomarker of Postexertional Malaise in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Rayhan RU, Baraniuk JN. Front Neurosci. 2021 Dec 15;15:748426. https://doi.org/10.3389/fnins.2021.748426
Conclusion/Main finding: «The dynamic increase in activation of the anterior DMN node after exercise may be a biomarker of postexertional malaise and symptom exacerbation in CFS. The specificity of this postexertional finding in ME/CFS can now be assessed by comparison to post-COVID fatigue, Gulf War Illness, fibromyalgia, chronic idiopathic fatigue, and fatigue in systemic medical and psychiatric diseases.»
Inclusion criteria: Fukuda/CDC and Canada Consensus Criteria
30. The Prospects of the Two-Day Cardiopulmonary Exercise Test (CPET) in ME/CFS Patients: A Meta-Analysis. Lim EJ, Kang EB, Jang ES, Son CG. J Clin Med. 2020 Dec 14;9(12):4040. doi: 10.3390/jcm9124040. PMID: 33327624; PMCID: PMC7765094.https://pubmed.ncbi.nlm.nih.gov/33327624/
Conclusion/Main finding: «The overall mean values of all parameters were lower on the 2nd day of the CPET than the 1st in ME/CFS patients, while it increased in the controls. From the meta-analysis, the difference between patients and controls was highly significant at Workload@VT (overall mean: -10.8 at test 1 vs. -33.0 at test 2, p<0.05), which may reflect present the functional impairment associated with PEM.».
Inclusion criteria: Metaanalysis
29. Comparing Idiopathic Chronic Fatigue and Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) in Males: Response to Two-Day Cardiopulmonary Exercise Testing Protocol C. (Linda) M. C. van Campen and Frans C. Visser, Healthcare 2021, 9(6), 683; https://doi.org/10.3390/healthcare9060683
Conclusion/Main finding: «This study confirms that male ME/CFS patients have a reduction in exercise capacity in response to a second-day CPET. These results are similar to published results in male ME/CFS populations. Patients diagnosed with ICF show a different response on day 2, more similar to sedentary and healthy controls.».
Inclusion criteria: International Consensus Criteria
28. Deconditioning does not explain orthostatic intolerance in ME/CFS (myalgic encephalomyelitis/chronic fatigue syndrome) van Campen, C. (Linda) M.C., Rowe, P.C. & Visser, F.C. J Transl Med 19, 193 (2021). https://doi.org/10.1186/s12967-021-02819-0
Conclusion/Main finding: «This study shows that in ME/CFS patients’ orthostatic intolerance is not caused by deconditioning as defined on cardiopulmonary exercise testing. An abnormal high decline in cerebral blood flow during orthostatic stress was present in all ME/CFS patients regardless of their % peak VO2 results on cardiopulmonary exercise testing.».
Inclusion criteria: International Consensus Criteria
27. Insights from Invasive Cardiopulmonary Exercise Testing of Patients with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (not two-day test!) Phillip Joseph, MD, Carlo Arevalo, MD, Rudolf K.F. Oliveira, MD, PhD, Mariana Faria- Urbina, MD, Donna Felsenstein, MD, Anne Louise Oaklander, MD, PhD, David M. Systrom, MD PII: S0012-3692(21)00256-7, DOI: https://doi.org/10.1016/j.chest.2021.01.082
Conclusion/Main finding: «These results identify two types of peripheral neurovascular dysregulation that are biologically plausible contributors to ME/CFS exertional intolerance—depressed Qc [Cardiac output] from impaired venous return, and impaired peripheral oxygen extraction. In patients with small-fiber pathology, neuropathic dysregulation causing microvascular dilation may limit exertion by shunting oxygenated blood from capillary beds and reducing cardiac return.».
Inclusion criteria: SEID criteria (acc. to IOM report 2015)
26. Exercise alters brain activation in Gulf War Illness and Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Stuart D Washington, Rakib U Rayhan, Richard Garner, Destie Provenzano, Kristina Zajur, Florencia Martinez Addiego, John W VanMeter, James N Baraniuk, Brain Communications, Volume 2, Issue 2, 2020, fcaa070, https://doi.org/10.1093/braincomms/fcaa070
Conclusion/Main finding: «Further, exercise caused increased activation among Myalgic Encephalomyelitis/Chronic Fatigue Syndrome patients within the dorsal midbrain, left operculo-insular cortex (Rolandic operculum) and right middle insula. … As they only emerge post-exercise, these regional differences likely represent neural substrates of cognitive post-exertional malaise useful for developing distinct diagnostic criteria for Gulf War Illness and Myalgic Encephalomyelitis/Chronic Fatigue Syndrome.»
Inclusion criteria: Fukuda/CDC
25. The Prospects of the Two-Day Cardiopulmonary Exercise Test (CPET) in ME/CFS Patients: A Meta Analysis Eun-Jin Lim, Eun-Bum Kang, Eun-Su Jang and Chang-Gue Son J. Clin. Med. 2020, 9(12), 4040; https://doi.org/10.3390/jcm9124040
Conclusion/Main finding: «The meta-analysis indicates a significant alteration of workload at VT especially on the 2nd day of CPET in ME/CFS patients. Accordingly, the two-day CPET could be considered as one of the potential objective assessment tools for PEM in ME/CFS patients.».
Inclusion criteria: Metaanalysis
24. Post-exertional symptoms distinguish Myalgic Encephalomyelitis/Chronic Fatigue Syndrome subjects from healthy controls Mateo, Lariel J.; Chu, Lily; Stevens, Staci; Stevens, Jared; Snell, Christopher R.; Davenport, Todd; VanNess, J. Mark Journal: Work, vol. 66, no. 2, pp. 265-275, 2020, DOI: 10.3233/WOR-203168 https://content.iospress.com/articles/work/wor203168
Conclusion/Main finding: «A standardized exertional stimulus produced prolonged, diverse symptoms in ME/CFS subjects. This provides clues to the underlying pathophysiology of ME/CFS, leading to improved diagnosis and treatment.».
Inclusion criteria: Fifteen of the 49 were formally recruited for the study and met the Fukuda (1994) criteria. The remaining 34 were physician diagnosed.
23. Validity of 2-Day Cardiopulmonary Exercise Testing in Male Patients with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome, van Campen, C. (Linda) M. C.; Rowe, Peter C.; Visser, Frans C. (2020). Advances in Physical Education. 10 (01): 68–80. DOI: 10.4236/ape.2020.101007 . ISSN 2164-0386.
Conclusion/Main finding: «The larger sample size of this study improves the confidence with which we can conclude that, like females, males have a similar decrement on day 2 of the consecutive day exercise tests. Our results confirm that 2-day CPET can be used in males to demonstrate the decrease in exercise capacity in research studies and if needed for social security claims. Further comparisons are needed to explore whether the absolute or relative changes in VO2 and workload on day 2 versus day 1 are similar across a wider range of clinical severity, and whether these values differ for subgroups with specific comorbid conditions.».
Inclusion criteria: Fukuda/CDC and International Consensus Criteria
22. Two-Day Cardiopulmonary Exercise Testing in Females with a Severe Grade of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Comparison with Patients with Mild and Moderate Disease C (Linda) MC van Campen, Peter C. Rowe, Frans C. Visser Healthcare 2020, 8(3), 192; doi:10.3390/healthcare8030192 https://www.mdpi.com/2227-9032/8/3/192
Conclusion/Main finding: «This is the first study to demonstrate that disease severity negatively influences exercise capacity in female ME/CFS patients. Finally, this study shows that the deterioration in peak workload from day-1 to day-2 is largest in the severe ME/CFS patient group.».
Inclusion criteria: Fukuda/CDC and International Consensus Criteria
21. Properties of measurements obtained during cardiopulmonary exercise testing in individuals with myalgic encephalomyelitis/chronic fatigue syndrome. Davenport TE, Stevens SR, Stevens MAJ, Snell CR, Van Ness JM.Work. 2020 Jun 16. doi: 10.3233/WOR-203170. Online ahead of print.PMID: 32568145 https://pubmed.ncbi.nlm.nih.gov/32568145/
Conclusion/Main finding: «CPET measurements demonstrated moderate to high reliability for individuals with ME/CFS. Comparing subjects with ME/CFS and control subjects yielded moderate to large effect sizes on all CPET measurements. MDC95 for all individuals with ME/CFS generally exceeded control subjects and CoVs for CPET measurements were comparable between groups.».
Inclusion criteria: Fukuda/CDC and International Consensus Criteria
20. A Machine Learning Approach to the Differentiation of Functional Magnetic Resonance Imaging Data of Chronic Fatigue Syndrome (CFS) From a Sedentary Control Destie Provenzano, Stuart D. Washington and James N. Baraniuk Front. Comput. Neurosci., 29 January 2020 https://doi.org/10.3389/fncom.2020.00002
Conclusion/Main finding: «The logistic regression model performed on fMRI data significantly differentiated CFS from control with model accuracy of 80.9% on Day 1 before exercise and 76.1% on Day 2 during the period of post-exertional malaise.».
Inclusion criteria: Fukuda/CDC, but argues for using Canada Consensus Criteria
19. Elevated blood lactate in resting conditions correlate with post-exertional malaise severity in patients with Myalgic encephalomyelitis/Chronic fatigue syndrome Alaa Ghali, Carole Lacout, Maria Ghali, Aline Gury, Anne-Berengere Beucher, Pierre Lozac’h, Christian Lavigne & Geoffrey Urbanski, Sci Rep. 2019 Dec 11;9(1):18817. https://doi.org/10.1038/s41598-019-55473-4.
Conclusion/Main finding: «ME/CFS patients with elevated blood lactate at rest may be at higher risk for more severe PEM.».
Inclusion criteria: International Consensus Criteria
18. Unexplained exertional intolerance associated with impaired systemic oxygen extraction Melamed KH, Santos M, Oliveira RKF, Urbina MF, Felsenstein D, Opotowsky AR, Waxman AB, Systrom DM. Eur J Appl Physiol. 2019 Sep 6. doi: 10.1007/s00421-019-04222-6. PMID: 31493035. https://www.ncbi.nlm.nih.gov/pubmed/31493035
Conclusion/Main finding: «We identified a cohort of patients whose exercise limitation is due only to systemic oxygen extraction, due to either an intrinsic abnormality of skeletal muscle mitochondrion, limb muscle microcirculatory dysregulation, or hyperventilation and left shift the oxyhemoglobin dissociation curve.».
Inclusion criteria: Own. Unexplained exertional intolerance, including those with suspected mitochondrial disease, were analyzed.
More about the study: Health Rising (2019): Poor Oxygen Extraction is Contributing to Exercise Intolerance in Chronic Fatigue Syndrome (ME/CFS)
17. Reproducibility of Measurements Obtained During Cardiopulmonary Exercise Testing in Individuals With Fatiguing Health Conditions – A Case Series Larson B, Davenport TE, Stevens SR, Stevens J, Van Ness JM, Snell CR Cardiopulmonary Physical Therapy Journal: June 24, 2019 – Volume Publish Ahead of Print – Issue – p doi: 10.1097/CPT.0000000000000100.
Conclusion/Main finding: «Nondisabled clients and clients with MS and HIV reproduced or improved in their volume of oxygen consumed (VO2), workload (WL), heart rate (HR), and minute ventilation (VE) at ventilatory anaerobic threshold (VAT) and at peak exercise (except peak WL and VE for the individual with HIV). Neither individual with ME/CFS reproduced VO2, WL, HR, or VE at VAT within literature estimates.».
Inclusion criteria: Canada Consensus Criteria
16. Abnormal blood lactate accumulation during repeated exercise testing in myalgic encephalomyelitis/chronic fatigue syndrome. Lien K, Johansen B, Veierød MB, Haslestad AS, Bøhn SK, Melsom MN, Kardel KR, Iversen PO. Physiol Rep. 2019 Jun;7(11):e14138. doi: 10.14814/phy2.14138. PMID: 31161646; PMCID: PMC6546966. https://www.ncbi.nlm.nih.gov/pubmed/31161646
Conclusion/Main finding: «In conclusion, previous exercise deteriorates physical performance and increases [Laa] during exercise in patients with ME/CFS while it lowers [Laa] in healthy subjects.».
Inclusion criteria: Canada Consensus Criteria
15. Chronotropic Intolerance: An Overlooked Determinant of Symptoms and Activity Limitation in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome? Davenport TE, Lehnen M, Stevens SR, VanNess JM, Stevens J, Snell CR. Front Pediatr. 2019 Mar 22;7:82. doi: 10.3389/fped.2019.00082. eCollection 2019. Review. PMID: 30968005; PMCID: PMC6439478. https://www.ncbi.nlm.nih.gov/pubmed/30968005
Conclusion/Main finding: «This literature synthesis supports the presence of abnormally blunted HR responses to activity in people with ME/CFS, at both maximal exertion and submaximal VAT.».
Inclusion criteria: Metaanalysis
14. Whole blood human transcriptome and virome analysis of ME/CFS patients experiencing post exertional malaise following cardiopulmonary exercise testing. Bouquet J, Li T, Gardy JL, Kang X, Stevens S, Stevens J, VanNess M, Snell C, Potts J, Miller RR, Morshed M, McCabe M, Parker S, Uyaguari M, Tang P, Steiner T, Chan WS, De Souza AM, Mattman A, Patrick DM, Chiu CY. PLoS One. 2019 Mar 21;14(3):e0212193. doi: 10.1371/journal.pone.0212193. eCollection 2019. PMID: 30897114; PMCID: PMC6428308. https://www.ncbi.nlm.nih.gov/pubmed/30897114
Conclusion/Main finding: «Although ME/CFS patients showed significant worsening of symptoms following exercise versus controls, with 8 of 14 ME/CFS patients showing reduced oxygen consumption (VO2) on day 2, transcriptome analysis yielded only 6 differentially expressed gene (DEG) candidates when comparing ME/CFS patients to controls across all time points.».
Inclusion criteria: Canada Consensus Criteria
13. Diagnostic sensitivity of 2-day cardiopulmonary exercise testing in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Nelson MJ, Buckley JD, Thomson RL, Clark D, Kwiatek R, Davison K. J Transl Med. 2019 Mar 14;17(1):80. doi: 10.1186/s12967-019-1836-0. PMID: 30871578; PMCID: PMC6417168. https://www.ncbi.nlm.nih.gov/pubmed/30871578
Conclusion/Main finding: «The decrease in WR (work rate) at VT (ventilatory threshold) of 6.3-9.8% on the 2nd day of consecutive-day CPET may represent an objective biomarker that can be used to assist with the diagnosis of ME/CFS.».
Inclusion criteria: Fukuda/CDC or Canada Consensus Criteria or International Consensus Criteria
12. Cardiopulmonary Exercise Test Methodology for Assessing Exertion Intolerance in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Stevens S, Snell C, Stevens J, Keller B, VanNess JM. Front Pediatr. 2018 Sep 4;6:242. doi: 10.3389/fped.2018.00242. eCollection 2018. PMID: 30234078; PMCID: PMC6131594. https://www.ncbi.nlm.nih.gov/pubmed/30234078
Conclusion/Main finding: «The second CPET measures changes in energy production and physiological function, objectively documenting the effects of post-exertional malaise.».
Inclusion criteria: SEID criteria (acc. to IOM report 2015) and International Consensus Criteria
11. Physiological measures in participants with chronic fatigue syndrome, multiple sclerosis and healthy controls following repeated exercise: a pilot study. Hodges LD, Nielsen T, Baken D. Clin Physiol Funct Imaging. 2018 Jul;38(4):639-644. doi: 10.1111/cpf.12460. Epub 2017 Aug 7. PMID: 28782878. https://www.ncbi.nlm.nih.gov/pubmed/28782878
Conclusion/Main finding: «These results suggest that exercise exhibits a different physiological response in MS and CFS/ME, demonstrating repeated cardiovascular exercise testing as a valid measure for differentiating between fatigue conditions.».
Inclusion criteria: Fukuda/CDC and Canada Consensus Criteria and International Consensus Criteria
10. A Pair of Identical Twins Discordant for Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Differ in Physiological Parameters and Gut Microbiome Composition Giloteaux, Ludovic; Hanson, Maureen R.; Keller, Betsy A., American Journal of Case Reports. 17: 720–729. doi:10.12659/AJCR.900314. ISSN 1941-5923. PMC 5058431 Freely accessible. PMID 27721367. (Oct 10, 2016). https://www.amjcaserep.com/abstract/index/idArt/900314
Conclusion/Main finding: «Results suggest dysfunctional immune activation in ILL following exercise and that prokaryotic viruses may contribute to mucosal inflammation and bacterial dysbiosis. Therefore, a two-day CPET and molecular analyses of blood and microbiomes could provide valuable information about ME/CFS, particularly if applied to a larger cohort of monozygotic twins».
Inclusion criteria: Unknown, but PEM is mentioned
9. Changes in Gut and Plasma Microbiome following Exercise Challenge in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS). Shukla SK, Cook D, Meyer J, Vernon SD, Le T, Clevidence D, Robertson CE, Schrodi SJ, Yale S, Frank DN. PLoS One. 2015 Dec 18;10(12):e0145453. doi: 10.1371/journal.pone.0145453. eCollection 2015. PMID: 26683192; PMCID: PMC4684203. https://www.ncbi.nlm.nih.gov/pubmed/26683192
Conclusion/Main finding: «Upon exercise challenge, there were significant changes in the abundance of major bacterial phyla in the gut in ME/CFS patients not observed in healthy controls. In addition, compared to controls clearance of bacteria from the blood was delayed in ME/CFS patients following exercise.».
Inclusion criteria: Fukuda/CDC
8. Inability of myalgic encephalomyelitis/chronic fatigue syndrome patients to reproduce VO₂peak indicates functional impairment. Keller BA, Pryor JL, Giloteaux L. J Transl Med. 2014 Apr 23;12:104. doi: 10.1186/1479-5876-12-104. PMID: 24755065; PMCID: PMC4004422. https://www.ncbi.nlm.nih.gov/pubmed/24755065
Conclusion/Main finding: «ME/CFS participants were unable to reproduce most physiological measures at both maximal and ventilatory threshold intensities during a CPET performed 24 hours after a prior maximal exercise test.».
Inclusion criteria: Fukuda/CDC
7. Decreased oxygen extraction during cardiopulmonary exercise test in patients with chronic fatigue syndrome. Vermeulen RC, Vermeulen van Eck IW. J Transl Med. 2014 Jan 23;12:20. doi:10.1186/1479-5876-12-20. PMID: 24456560; PMCID: PMC3903040. https://www.ncbi.nlm.nih.gov/pubmed/24456560
Conclusion/Main finding: «Low oxygen uptake by muscle cells causes exercise intolerance in a majority of CFS patients, indicating insufficient metabolic adaptation to incremental exercise. The high increase of the cardiac output relative to the increase of oxygen uptake argues against deconditioning as a cause for physical impairment in these patients.».
Inclusion criteria: Fukuda/CDC
6. Discriminative validity of metabolic and workload measurements for identifying people with chronic fatigue syndrome. Snell CR, Stevens SR, Davenport TE, Van Ness JM. Phys Ther. 2013 Nov;93(11):1484-92. doi:10.2522/ptj.20110368. Epub 2013 Jun 27. PMID: 23813081. https://www.ncbi.nlm.nih.gov/pubmed/23813081
Conclusion/Main finding: «Multivariate analysis showed no significant differences between control participants and participants with CFS for test 1. However, for test 2, participants with CFS achieved significantly lower values for oxygen consumption and workload at peak exercise and at the ventilatory or anaerobic threshold. Follow-up classification analysis differentiated between groups with an overall accuracy of 95.1%.».
Inclusion criteria: Fukuda/CDC but PEM required
5. Diagnostic accuracy of symptoms characterising chronic fatigue syndrome. Todd E. Davenport, Staci R. Stevens, Katie Baroni, Mark Van Ness & Christopher R. Snell, (2011), Disability and Rehabilitation, 33:19-20, 1768-1775, DOI: 10.3109/09638288.2010.546936
Conclusion/Main finding: «A cluster of associated symptoms distinguishes between individuals with and without CFS. Fewer associated symptoms may be necessary to establish a diagnosis of CFS than currently described.»
Inclusion criteria: Fukuda/CDC
4. Patients with chronic fatigue syndrome performed worse than controls in a controlled repeated exercise study despite a normal oxidative phosphorylation capacity. Vermeulen RC, Kurk RM, Visser FC, Sluiter W, Scholte HR. J Transl Med. 2010 Oct 11;8:93. doi:10.1186/1479-5876-8-93. PMID: 20937116; PMCID: PMC2964609. https://www.ncbi.nlm.nih.gov/pubmed/20937116
Conclusion/Main finding: «At both exercise tests the patients reached the anaerobic threshold and the maximal exercise at a much lower oxygen consumption than the controls and this worsened in the second test. This implies an increase of lactate, the product of anaerobic glycolysis, and a decrease of the mitochondrial ATP production in the patients.».
Inclusion criteria: Fukuda/CDC as consequence of infection
3. Postexertional malaise in women with chronic fatigue syndrome. VanNess JM, Stevens SR, Bateman L, Stiles TL, Snell CR. J Womens Health (Larchmt). 2010 Feb;19(2):239-44. doi: 10.1089/jwh.2009.1507. PMID: 20095909. https://www.ncbi.nlm.nih.gov/pubmed/20095909
Conclusion/Main finding: «The results of this study suggest that PEM is both a real and an incapacitating condition for women with CFS and that their responses to exercise are distinctively different from those of sedentary controls.».
Inclusion criteria: Fukuda/CDC
2. Diminished Cardiopulmonary Capacity During Post-Exertional Malaise. Vanness, J. Mark; Snell, Christopher R.; Stevens, Staci R. Journal of Chronic Fatigue Syndrome.14 (2): 77–85. doi:10.1300/j092v14n02_07. ISSN 1057-321. 2007. https://www.tandfonline.com/doi/abs/10.1300/J092v14n02_07
Conclusion/Main finding: «In the absence of a second exercise test, the lack of any significant differences for the first test would appear to suggest no functional impairment in CFS patients. However, the results from the second test indicate the presence of a CFS related post-exertional malaise. It might be concluded then that a single exercise test is insufficient to demonstrate functional impairment in CFS patients. A second test may be necessary to document the atypical recovery response and protracted malaise unique to CFS.».
Inclusion criteria: Fukuda/CDC
1. Chronic fatigue syndrome: new evidence for a central fatigue disorder. Georgiades E, Behan WM, Kilduff LP, Hadjicharalambous M, Mackie EE, Wilson J, Ward SA, Pitsiladis YP. Clin Sci (Lond). 2003 Aug;105(2):213-8. doi: 10.1042/CS20020354. PMID: 12708966. https://www.ncbi.nlm.nih.gov/pubmed/12708966
Conclusion/Main finding: « the significant differences between patients with CFS and healthy controls that we observed in several key CNS 5-HT and dopaminergic modulators, assuming that they are indeed reflective of brain 5-HT and dopamine levels, suggest that central neural mechanisms may contribute to the increased perception of effort and impaired exercise tolerance in CFS.».
Inclusion criteria: Fukuda/CDC
Summaries
D. Lessons from Myalgic Encephalomyelitis/Chronic Fatigue Syndrome for Long COVID Part 4: Heart Rate Monitoring to Manage Postexertional Symptom Exacerbation Todd E. Davenport, Staci R. Stevens, Jared Stevens, Christopher R. Snell, J. Mark Van Ness Published online on February 23, 2022, https://www.jospt.org/do/10.2519/jospt.blog.20220223/full/
Conclusion/Main finding: «A combination of HRM and symptom journaling will best assist patients and clinicians to track outcomes and adjust the HRM program as needed. The patient and clinician should collaborate to determine the “Key 3” symptoms of PESE that are most functionally limiting to the patient. Symptoms may be assessed using a formalized questionnaire,1,3,9 or they may be qualitatively determined based on discussion with the patient. The presence and severity of Key 3 symptoms can be reassessed over time to determine the effectiveness of the HRM program.»
Inclusion criteria: Not applicable
C. Lessons from Myalgic Encephalomyelitis/Chronic Fatigue Syndrome for Long COVID Part 3: “Energy System First Aid” for People With Postexertional Symptom Exacerbation Todd E. Davenport, Staci R. Stevens, Jared Stevens, Christopher R. Snell, J. Mark Van Ness Published online on February 16, 2022, https://www.jospt.org/do/10.2519/jospt.blog.20220216/full/
Conclusion/Main finding: «First aid is an important part of rehabilitation. Energy system first aid for people with PESE focuses on helping patients to use the metabolic systems that function well, and to limit the use of the aerobic system that current evidence suggests is functioning abnormally. Working together, patients and clinicians can use widely available tools and simple insights from physiological data to promote improvements in symptoms and functioning»
Inclusion criteria: Not applicable
B. Lessons from Myalgic Encephalomyelitis/Chronic Fatigue Syndrome for Long COVID Part 2: Physiological Characteristics During Acute Exercise Are Abnormal in People With Postexertional Symptom Exacerbation Todd E. Davenport, Staci R. Stevens, Jared Stevens, Christopher R. Snell, J. Mark Van Ness Published online February 9, 2022, https://www.jospt.org/do/10.2519/jospt.blog.20220209/full/
Conclusion/Main finding: «This post reviewed evidence from systems-level physiology that indicates important differences in physiological responses to acute exercise between people with deconditioning compared to people with PESE. Clearly, PESE is not deconditioning. Rather, physiological changes may be related to autonomic dysfunction, as well as direct and indirect effects of pathogenic infection, such as the novel coronavirus infection in long COVID. Insights into the physiology of PESE may be used to evaluate and treat people with long COVID and ME/CFS, as well as shape future rehabilitation research and safe clinical practices.»
Inclusion criteria: Not applicable
A. Lessons from Myalgic Encephalomyelitis/Chronic Fatigue Syndrome for Long COVID: Postexertional Symptom Exacerbation is an Abnormal Response to Exercise/Activity Todd E. Davenport, Staci R. Stevens, Jared Stevens, Christopher R. Snell, J. Mark Van Ness Published online February 2, 2022. https://www.jospt.org/do/10.2519/jospt.blog.20220202/full/
Conclusion/Main finding: «Onset and duration of PESE vary for most people with ME/CFS. It is most common to begin presenting immediately after a triggering activity but may be delayed in ~11% of individuals.2 Failure to report recovery to baseline symptom levels and functioning within 24 hours following a serial CPET differentiates people with ME/CFS and sedentary individuals who experienced a normal postexercise recovery (11-fold increased likelihood).3 This observation suggests that an inability to recover from an exercise/activity challenge within a day is a diagnostically useful question for physical therapists and should be considered when designing the plan of care.»
Inclusion criteria: Not applicable
Other
4. The biggest 2-day exercise study. Blogpost by ME/CFS Skeptic posted on September 18th, 2024: https://mecfsskeptic.com/the-biggest-2-day-exercise-study/
3. “Post-Exertional Malaise (PEM): Systematic literature search” Norwegian Institute of Public Health, February 2019. Mapping of research on exertion-triggered illness/worsening of symptoms (Post-Exertional Malaise, PEM) in chronic fatigue syndrome CFS/ME. A systematic literature search has been performed, the references from the search have been reviewed and relevant references have been sorted into different categories. https://www.fhi.no/en/publ/2019/Post-Exertional-Malaise-PEM/ (Report in Norwegian only).
2. Summary of Exercise Provocation Studies in ME/CFS, Mary Dimmock, 2023
1. ME/CFS Pathophysiology Investigated by Invasive Cardiopulmonary Exercise Testing and Autonomic Function Testing, conference abstract, J. Squires, K. Wichmann Madsen, M.C. Stovall, S. Al-Zayer, W. Xiao, C.-J. Chang, P. Novak, and D.M. Systrom, B30. BREATHE (IN THE AIR): STUDIES IN PULMONARY FUNCTION. May 1, 2023, A2996-A2996
Videos and webinars that describes exercise tests and explains PEM
- Miriam E. Tucker “No Evidence Supports Using Graded Exercise for Myalgic Encephalomyelitis/Chronic Fatigue Syndrome”, 2025
Most studies examining the effects of activity-based interventions for myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) have not enrolled people who have the condition as it’s currently defined, requiring the core symptom of post-exertional malaise (PEM), a new analysis found.
https://www.medscape.com/viewarticle/no-evidence-supports-using-graded-exercise-myalgic-2025a1000tuf - Prof. Dr. Todd Davenport «Phenotyping Responses to Exertion in ME/CFS & Long Covid (Day 2, Block 6)» May 15th, 2024. https://youtu.be/YHu87Cbhj5o?si=jhrE3YwQ0aU8FTK
- Dr. Lucinda Bateman, MD, Dr. Brayden Yellman, MD. Bateman Horne Center, “Post-Exertional Malaise – Cardiopulmonary Exercise Studies” August 15th, 2023: https://fb.watch/mwmfUd-rX0/
- David M. Systrom, MD, Director of the Massachusetts General Hospital Cardiopulmonary laboratory, Boston, Massachusetts. Assistant Professor of Medicine at Harvard Medical School: “ME/CFS and Long Covid: Insights From Invasive Cardiopulmonary Exercise Testing” June 29th, 2023.: https://youtu.be/SeW2AA0bc2Y
- Clayton Powers, DPT, “Therapy for patients with PEM, When exercise causes harm” July 5th, 2023: https://youtu.be/ioSe6LPVLDM
- Todd Davenport, professor of physical therapy at University of the Pacific in Stockton, California, interviewed by David Tuller, DrPH: “Trial By Error: Todd Davenport on Post-Exertional Symptom Exacerbation in Long Covid and ME/CFS”, August 22nd, 2022: https://www.virology.ws/2022/08/22/trial-by-error-todd-davenport-on-post-exertional-symptom-exacerbation-in-long-covid-and-me-cfs-2/
- David M. Systrom, MD, Director of the Massachusetts General Hospital Cardiopulmonary laboratory, Boston, Massachusetts. Assistant Professor of Medicine at Harvard Medical School “Pathophysiology of Exercise Intolerance in Chronic Fatigue Syndrome” 24 October, 2020: https://youtu.be/TyQpilazB_E
- Professor Christopher R. Snell, Pacific Fatigue Laboratory, Calefornia, USA, “ME/CFS, CPET, PEM, COVID, GET and more” 28. august, 2020: https://m.youtube.com/watch?v=2fJt14pJymw
- David M. Systrom, MD, Director of the Massachusetts General Hospital Cardiopulmonary laboratory, Boston, Massachusetts. Assistant Professor of Medicine at Harvard Medical School “Pathophysiology of Exercise Intolerance in Chronic Fatigue Syndrome” May 5th, 2020: https://youtu.be/Q335NFTSZno
- Staci Stevens, Todd Davenport and Mark VanNess. “Why Working out Doesn’t Work. Answers from 20 years of cardiopulmonary exercise testing (CPET) in ME/CFS” December 18th, 2019: https://vimeo.com/380338703
- Professor Betsy Keller, PhD, Ithaca College, New York “PEM – ergospirometry testing and coping strategies to avoid PEM”, December 8th, 2019 (Norwegian subtitles): https://youtu.be/nzY_7Z-eUcA
- Staci Stevens, Dr. Chris Snell, Mark VanNess, Brian Hughes, Jonathan Edwards “Understanding Graded Exercise Therapy for ME/CFS” – built on a false premise” November 1st, 2019: https://www.dialogues-mecfs.co.uk/films/graded-exercise-therapy/
- David M. Systrom, MD, Director of the Massachusetts General Hospital Cardiopulmonary laboratory, Boston, Massachusetts. Assistant Professor of Medicine at Harvard Medical School “Pathophysiology and treatment of exertional intolerance in ME/CFS: insights from cardiopulmonary exercise testing” April 4th, 2019: https://youtu.be/1emsA2CcRK4?t=17636
- Katarina Lien, MD and PhD student, University of Oslo “Post Exertional Malaise in ME/CFS patients” January 14th, 2019 (English subtitle): https://youtu.be/ol96vxqJ8rA
- David M. Systrom, MD, Director of the Massachusetts General Hospital Cardiopulmonary laboratory, Boston, Massachusetts. Assistant Professor of Medicine at Harvard Medical School “Advancements in ME/CFS Research”, David M. Systrom, MD; Brigham and Women’s Hospital | ME/CFSAlert 98” July 2nd, 2018: https://m.youtube.com/watch?feature=youtu.be&v=FMaKfv8peww
- Dr. Peter Rowe, professor in paediatrics, John Hopkins hospital, USA, “Inducing Post Exertional Malaise: A look at the research evidence” July 16th, 2015: https://youtu.be/ux93w7yGQ5g
- Dr. Lily Chu, Stanford University, USA, “Post Exertional Malaise: History, Characteristics, Evidence” June 23rd, 2015: https://youtu.be/hxJPrkWHcBo
- Professor Christopher R. Snell, Pacific Fatigue Laboratory, California, USA, “Making the case for clinical exercise testing in CFS/ME research and treatment” Sept. 23rd, 2012: https://m.youtube.com/watch?v=nL49DwGRs30&t=1s
- Staci Stevens, MA in exercise physiology. Director of the Pacific Fatigue Lab, Founder and Director of Workwell Foundation, Ripon, California, USA, “MECFS Alert Episode 32“, August 6th, 2012: https://m.youtube.com/watch?feature=youtu.be&v=kCO3pAbSq3I
Patient experiences
• Interview with psychologist specialist and former professional cyclist Ingunn Ullerhaug EKKO, NRK Radio June 6th, 2019 (Norwegian speech only) https://radio.nrk.no/serie/ekko/MDSP25011219/06-06-2019#t=21m40.08s
• «From Tour de France to ME» Chronicle by psychologist specialist and former professional cyclist Ingunn Ullerhaug, NRK, June 6th, 2019 (Norwegian text) https://www.nrk.no/ytring/fra-tour-de-france-til-me-1.14611826
Written by
Jørn Tore Haugen
Master of Science in Engineering
Updated: 8th January 2022, 24th February 2022, 17th August 2022, 27th November 2022, 16th March 2023 and 18th June 2023, 17th January 2024, 2nd June 2025, October 16th, 2025, November 16th 2025 and January 26th 2026
Tilbaketråkk: NICE, Media & Statements – Life with ME by Sissel
Tilbaketråkk: 24 fagfolk opprettholder myter om ME | MElivet
Tilbaketråkk: Diagnosekriterier betyr noe! – Life with ME by Sissel
Tilbaketråkk: NICE gir oss håp! – Life with ME by Sissel
Tilbaketråkk: ME-livets årskavalkade 2021 | MElivet
Tilbaketråkk: Informasjonspakke – ME – Life with ME by Sissel
Tilbaketråkk: Doktorgradsavhandling sår tvil om ME | MElivet
Tilbaketråkk: Doctoral dissertation casts doubt on ME | MElivet
Tilbaketråkk: LP-studien & media – Life with ME by Sissel
Tilbaketråkk: Feil om ME – igjen | MElivet
Tilbaketråkk: Postkortaksjon 2022 – Life with ME by Sissel
Tilbaketråkk: Vil LP-studien ta hensyn til fysisk forverring? – Life with ME by Sissel
Tilbaketråkk: 10-års jubileum! | MElivet
Tilbaketråkk: WHO fraråder trening ved PEM! – Life with ME by Sissel
Tilbaketråkk: Morgenbladet polariserer ME-debatten – Life with ME by Sissel
Tilbaketråkk: Vil FHI slutte fred med ME-syke? – Life with ME by Sissel
Tilbaketråkk: Kvinnehelseutvalgets rapport er en skandale for ME-syke | MElivet
Tilbaketråkk: ME-forskere og FHI svikter ME-syke | MElivet
Tilbaketråkk: Elendig kvalitet på ME-forskning | MElivet
Tilbaketråkk: Poor quality of ME research | MElivet
Tilbaketråkk: Kvinnehelserapporten: Misvisende, mangelfull og partisk – Life with ME by Sissel
Tilbaketråkk: Mye skrik og lite ull i ME-debatten | MElivet
Tilbaketråkk: Danmark: Kontroversiel behandling af ME-patienter | MElivet
Tilbaketråkk: Uansvarlig av Nettavisen – Life with ME by Sissel
Tilbaketråkk: Esther Crawley MAGENTA studie for barn og unge med Myalgisk Encefalomyelitt ME, sier så mye mer om flere behandlingsterapier som ikke virker – NeuroImmunologisk Myalgisk Encefalomyelitt (ME)
Tilbaketråkk: Vi blir sykere av behandlingen deres. Hva forventer de? Takknemlighet? | MElivet
Tilbaketråkk: PEM – en lunefull fiende | Diplodokus
Tilbaketråkk: We deteriorate by their treatment. What do they expect? Gratitude? – The ME Global Chronicle
Tilbaketråkk: Etisk og vitenskapelig problematisk Lightning Process-studie – Life with ME by Sissel
Tilbaketråkk: Wyller på dypt vann – Life with ME by Sissel
Tilbaketråkk: Samvalg for pasienter med CFS/ME: Informasjon kan føre til sykdomsforverring | MElivet
Tilbaketråkk: Silje Reme fikk gi innspill likevel – Life with ME by Sissel
Tilbaketråkk: ME-pasienters brukermedvirkning – del 2 – Er ME-pasienter syke i sjelen? | MElivet
Tilbaketråkk: En kompetenshöjning måste baseras på erkänd forskning | MElivet
Tilbaketråkk: Kompetansetjenestens løse påstander – I | MElivet
Tilbaketråkk: Kompetansetjenestens løse påstander – II | MElivet
Tilbaketråkk: Kompetansetjenesten løse påstander – III | MElivet
Tilbaketråkk: ¿Por qué fallan las mitocondrias en EM/SFC y covid persistente? - Web informativa de Síndrome de Fatiga Crónica/ Encefalomielitis Miálgica y COVID persistente
Tilbaketråkk: Vetenskap ersätts av anekdoter i replik i Läkartidningen – The ME Inquiry Report
Tilbaketråkk: ME må ikke forveksles med helseangst | MElivet
Tilbaketråkk: Kunnskapsgrunnlaget V | MElivet
Tilbaketråkk: Høringsinnspill: Retningslinje for langvarig utmattelse, inkl. ME/CFS | MElivet
Tilbaketråkk: KI-oppsummering av 2-dagers CPET-studier | MElivet